1. Warth, B., et al. (2022).
“Mycotoxin-Linked Mutations and Cancer Risk: A Global Health Issue.” Frontiers in Oncology, 12, 863478. https://doi.org/10.3390/ijerph19137754
Study Type / Design: Comprehensive mechanistic and molecular review synthesizing human epidemiological, genomic, and toxicologic data on mycotoxin-induced carcinogenesis.
Population & Exposure Type:
Global human populations are chronically exposed to mycotoxins (primarily aflatoxins, ochratoxin A, fumonisins, trichothecenes) through food or contaminated indoor environments.
Main Findings (Human Cancer Links):
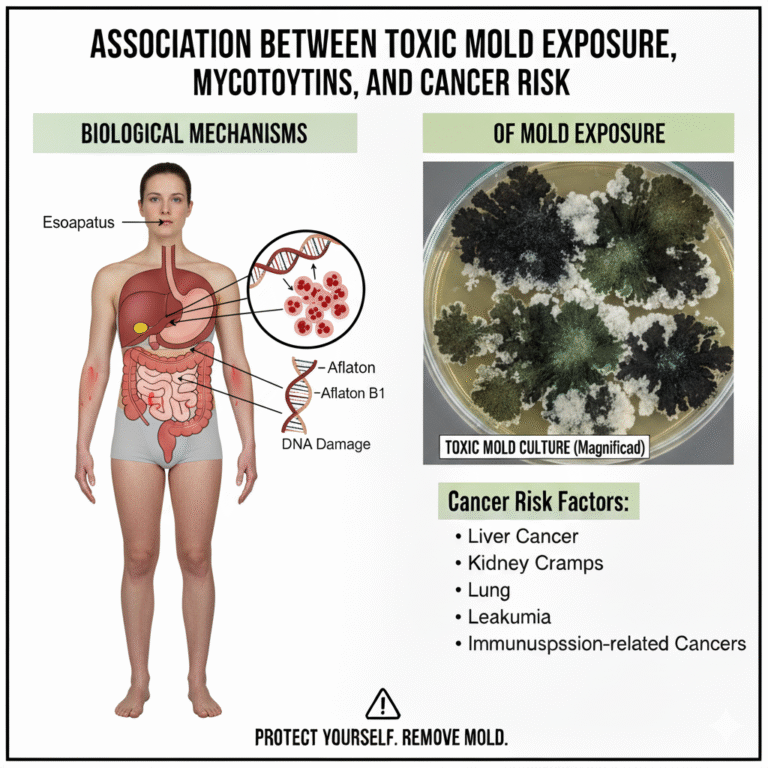
- Aflatoxin B₁ exposure causes a signature p53 codon 249 ser→arg mutation found in ≈ 30–60 % of hepatocellular carcinoma (HCC) cases in high-exposure regions (e.g., China, West Africa).
- Ochratoxin A (OTA) is associated with urothelial and renal tumors; detected in the serum and urine of affected patients.
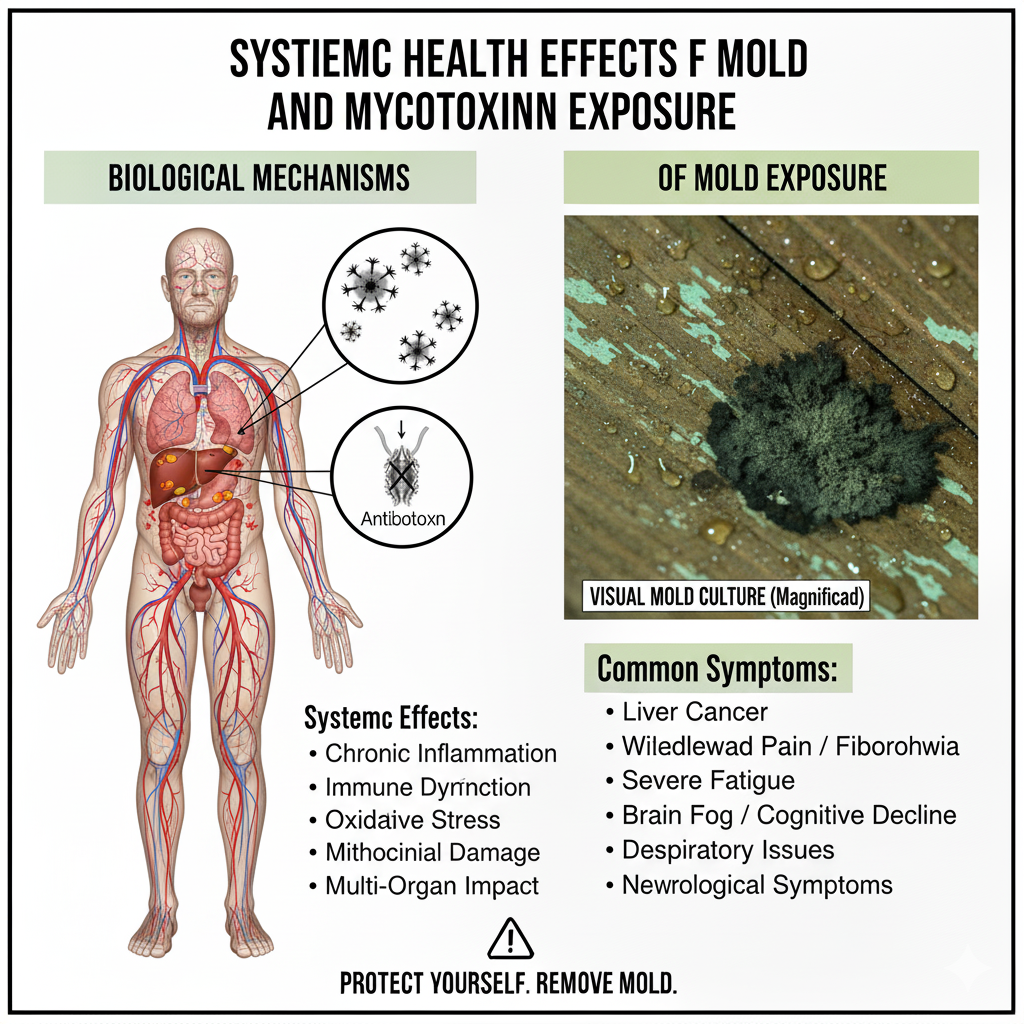
- Discusses emerging data on indoor mycotoxins (e.g., trichothecenes, satratoxins) as genotoxic and potentially co-carcinogenic via oxidative stress and DNA adducts.
- Emphasizes the need for genomic surveillance and global mitigation of mycotoxin exposure.
Effect Size / Strength:
For aflatoxin–HCC, the combined relative risk ≈ 6–30×, depending on HBV status (cited from prior meta-analyses). Other mold toxins: mechanistic but not yet quantified epidemiologically.
Causation Level:
- Definitive causal for aflatoxin B₁ → liver cancer (IARC Group 1).
- Probable cause for ochratoxin A → urothelial/renal tumors.
- Mechanistically suggestive for other indoor mold toxins (needs human validation).
Notes / Limitations:
Focuses mainly on dietary and environmental toxin pathways; limited direct indoor-mold epidemiology.
Quote::
“Aflatoxin B₁ exposure induces the hotspot p53 codon 249 mutation, a molecular signature in hepatocellular carcinoma, while ochratoxin A and other mycotoxins show genotoxic potential implicating them as emerging global cancer risk factors.” — Warth et al., 2022, p. 863478.
2. Claeys, L., Romano, C., De Ruyck, K., et al. (2020).
“Mycotoxin Exposure and Human Cancer Risk: A Systematic Review of Epidemiological Studies.” Food and Chemical Toxicology, 146, 111702. https://core.ac.uk/download/pdf/323232763.pdf
Study Type / Design: Systematic review of 42 human epidemiologic studies evaluating cancer outcomes in relation to mycotoxin exposure.
Population & Exposure Type:
Global cohorts with measured or estimated exposure to aflatoxins, ochratoxin A, fumonisins, trichothecenes, and zearalenone, primarily via food but also occupational settings.
Main Findings (Human Cancer Links):
- Strong evidence: aflatoxin B₁ → primary liver cancer (HCC).
- Moderate evidence: ochratoxin A → urothelial/renal carcinomas.
- Limited/inconsistent evidence for fumonisins → esophageal cancer.
- Insufficient data for other molds (including indoor exposure).
- Highlights the synergistic effect between HBV infection and aflatoxin exposure on HCC risk.
Effect Size / Strength:
Pooled HCC relative risk ≈ 6–13× for high vs low aflatoxin exposure.
OTA studies: OR ≈ 2–4 for renal cancer, but heterogeneous.
Causation Level:
- Causal (IARC Group 1): aflatoxins → HCC.
- Probable (Group 2B): ochratoxin A → urothelial/renal tumors.
- Insufficient: other indoor molds/mycotoxins.
Notes / Limitations:
Most studies are dietary; indoor air exposure is not quantified. Nonetheless, provides the highest-quality synthesis of human carcinogenic evidence for mycotoxins overall.
Quote::
“Aflatoxin B₁ exposure is causally linked to hepatocellular carcinoma, while ochratoxin A is probably carcinogenic to humans; evidence for other mycotoxins remains limited or inconsistent.” — Claeys et al., 2020, p. 111702.